
Full text loading...
Symbiogenesis is the extremely rare, but permanent merger of two organisms from phylogenetically distant lineages into one radically more complex organism. Three examples are exceptionally important: intracellular enslavement by an early eukaryote of an α-proteobacterium by host protein insertion to make mitochondria; later conversion of a cyanobacterium into the first chloroplast, thereby forming kingdom Plantae; and secondary enslavement of a red alga to yield more complex membrane topology in the phagophototrophic kingdom Chromista. Two other cases involved independent acquisition of green-algal chloroplasts by ancestrally phagotrophic lineages, yielding chlorarachnean algae (phylum Cercozoa, within the chromist infrakingdom Rhizaria) and euglenophyte algae (phylum Euglenozoa, within the protozoan subkingdom Eozoa). Less radically, chloroplast replacement occurred within dinoflagellate Chromista by two symbiogeneses: Green-algal or haptophyte chloroplasts replaced ancestral peridinin-containing chloroplasts. These seven lineage mergers were all mediated by the evolution of novel modes of transmembrane protein import into the enslaved cell, allowing massive gene transfer from symbionts to host genomes.

Article metrics loading...

Full text loading...


Data & Media loading...
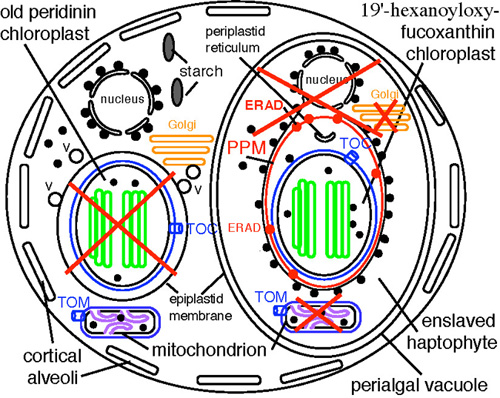
Supplemental Figure 1: Tertiary symbiogenesis generated the dinoflagellate family Kareniaceae by replacing its peridinin-containing chloroplast by a 19'-hexanoyloxy-fucoxanthin-containing one from a prymnesiophyte haptophyte. The haptophyte chloroplast was initially surrounded by six membranes if engulfed by phagocytosis or five if internalised by myzocytosis that removes prey plasma membranes. Now 2-4 only remain (number uncertain); all membranes marked by red crosses were eliminated. The simplest initial mode of enslavement involves haptophyte plasma membrane loss, periplastid ER breakage (remaining as vesicles), adding host Golgi vesicle targeting to the perialgal vacuole surrounding the haptophyte (either by fusing with host epiplastid membrane or with epiplastid-destined Golgi vesicles (V), and retaining haptophyte symbiont ERAD for PPM transit. Such reuse of pre-existing import machinery would make a chimaeric cell able to import chloroplast proteins across four membranes, perhaps still the condition in Karenia (Kite & Dodge 1985). Liberation into the cytosol by membrane breakage of a separate copy of the haptophyte nucleus and its fusion with the host nucleus would transfer all needed plastid protein genes with ready-made targeting sequences. Karenia retains both host and symbiont nucleus-encoded plastid proteins. If Karlodinium really has only two membranes (doubtful), the perialgal vacuole and PPM were lost and protein import uses only Toc/Tic machinery.
Kite GC, Dodge JD. 1985. Structural organization of plastid DNA in two anomalously pigmented dinoflagellates. J. Phycol. 21:50--56
Download Supplemental Figure 1 as a PDF.
