Multimedia
Filter :
Publication date
Article type
Subject
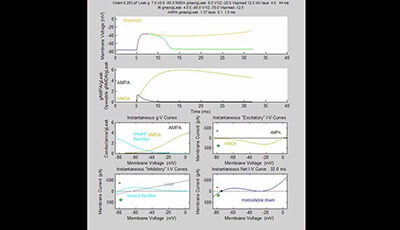
Active Properties of Neocortical Pyramidal Neuron Dendrites: Supplemental Video 3
A supplemental video from the 2013 review by Guy Major Matthew E. Larkum and Jackie Schiller "Active Properties of Neocortical Pyramidal Neuron Dendrites" from the Annual Review of Neuroscience.
The supplemental movies show time evolution of I-V curves and membrane potential (Vm) of models following a brief pulse of glutamate with AMPAR conductances yielding just subthreshold and just-suprathreshold responses. Pale orange line in top panel and X in bottom right panel indicate threshold when bistable state exists.
In Supplemental Videos 3 and 4 the highest gmax NMDA = 6 × leak. Video 3: gmax AMPA = 1.37 × leak (subthreshold). Video 4: gmax AMPA = 1.38 × leak (suprathreshold). Would see medium spike ‘riding on’ medium subthreshold response (if superimposed).
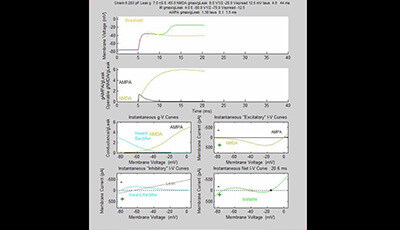
Active Properties of Neocortical Pyramidal Neuron Dendrites: Supplemental Video 4
A supplemental video from the 2013 review by Guy Major Matthew E. Larkum and Jackie Schiller "Active Properties of Neocortical Pyramidal Neuron Dendrites" from the Annual Review of Neuroscience.
The supplemental movies show time evolution of I-V curves and membrane potential (Vm) of models following a brief pulse of glutamate with AMPAR conductances yielding just subthreshold and just-suprathreshold responses. Pale orange line in top panel and X in bottom right panel indicate threshold when bistable state exists.
In Supplemental Videos 3 and 4 the highest gmax NMDA = 6 × leak. Video 3: gmax AMPA = 1.37 × leak (subthreshold). Video 4: gmax AMPA = 1.38 × leak (suprathreshold). Would see medium spike ‘riding on’ medium subthreshold response (if superimposed).
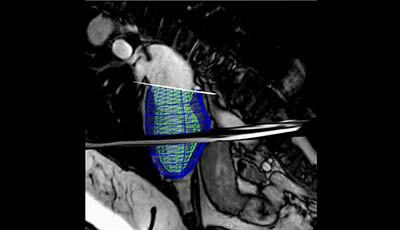
Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering: Figure 2
Figure 2 from the 2013 review by A.A. Young and J.L. Prince "Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering" from the Annual Review of Biomedical Engineering.
Video showing a 3D reconstruction of the left ventricle through the cardiac cycle.
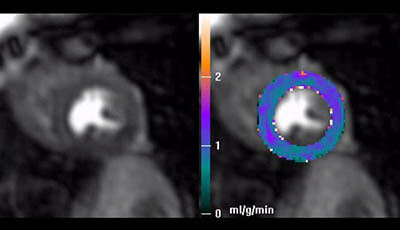
Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering: Figure 3: Rest
Figure 3 from the 2013 review by A.A. Young and J.L. Prince "Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering" from the Annual Review of Biomedical Engineering.
Video showing the time course of the perfusion scan capturing uptake of contrast by the myocardium over a period of ∼30 s and the perfusion parameters derived at rest (courtesy of: Hsu LY Groves DW Aletras AH Kellman P Arai AE. 2012. A quantitative pixel-wise measurement of myocardial blood flow by contrast-enhanced first-pass CMR perfusion imaging: microsphere validation in dogs and feasibility study in humans. JACC Cardiovasc. Imaging 5: 154–66).
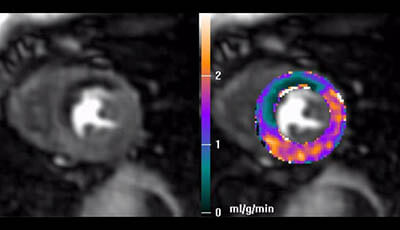
Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering: Figure 3: Stress
Figure 3 from the 2013 review by A.A. Young and J.L. Prince "Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering" from the Annual Review of Biomedical Engineering.
Video showing the time course of the perfusion scan capturing uptake of contrast by the myocardium over a period of ∼30 s and the perfusion parameters derived during adenosine stress (courtesy of: Hsu LY Groves DW Aletras AH Kellman P Arai AE. 2012. A quantitative pixel-wise measurement of myocardial blood flow by contrast-enhanced first-pass CMR perfusion imaging: microsphere validation in dogs and feasibility study in humans. JACC Cardiovasc. Imaging 5: 154–66).
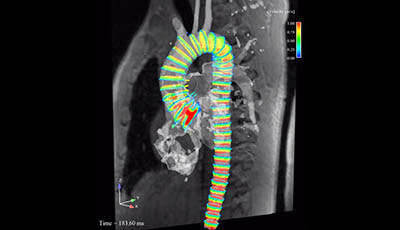
Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering: Figure 5
Figure 5 from the 2013 review by A.A. Young and J.L. Prince "Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering" from the Annual Review of Biomedical Engineering.
Video showing four-dimensional (4D) flow animation (courtesy of: Markl M Wallis W Brendecke S Simon J Frydrychowicz A Harloff A. 2010. Estimation of global aortic pulse wave velocity by flow-sensitive 4D MRI. Magn. Reson. Med. 63: 1575–82)
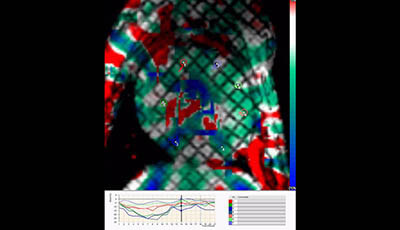
Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering: Figure 6
Figure 6 from the 2013 review by A.A. Young and J.L. Prince "Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering" from the Annual Review of Biomedical Engineering.
Video showing computer strain in a patient with an infarct (video courtesy of Donel Tani and Diagnosoft Inc.).
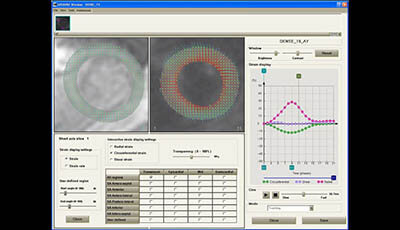
Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering: Figure 7
Figure 7 from the 2013 review by A.A. Young and J.L. Prince "Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering" from the Annual Review of Biomedical Engineering.
Video showing animation of dense displacements and circumferential strain.
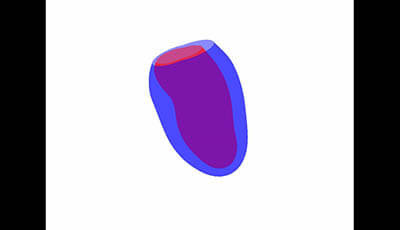
Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering: Figure 9: Upper 40 Phases
Figure 9 from the 2013 review by A.A. Young and J.L. Prince "Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering" from the Annual Review of Biomedical Engineering.
Video showing animation of plus two standard deviations in the most dominant spatiotemporal shape mode (courtesy of Pau Medrano-Gracia).
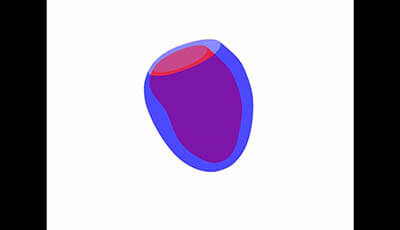
Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering: Figure 9: Lower 40 Phases
Figure 9 from the 2013 review by A.A. Young and J.L. Prince "Cardiovascular Magnetic Resonance: Deeper Insights Through Bioengineering" from the Annual Review of Biomedical Engineering.
Video showing animation of minus two standard deviations in the most dominant spatiotemporal shape mode (courtesy of Pau Medrano-Gracia).
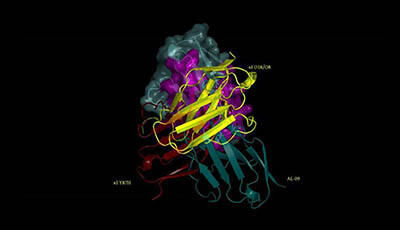
Systemic Amyloidoses: Video 1
Video 1 from the 2013 review by Luis M. Blancas-Mejía and Marina Ramirez-Alvarado "Systemic Amyloidoses" from the Annual Review of Biochemistry.
This morph animation shows the transition among three unique dimer orientations present in the crystal structures of κI O18/O8 and AL-09 as well as the nuclear magnetic resonance structure of κI Y87H to demonstrate the structural differences between them. This animation does not represent genuine conformational changes. The residues in monomer A involved in the κI dimer interface are in magenta whereas residues outside the dimer interface are in cyan. Monomer B (ribbons) of AL-09 (blue PDB code: 2Q1E) κI O18/O8 (gold PDB code: 2Q20) and κI Y87H (red PDB code: 2KQM) shows a ∼90° and ∼180° rotation with respect to the monomer A from the canonical dimer interface. In yellow ribbon are shown the differences in dimer structures like the hands on a clock moving in intervals of 90°. Interface residues Q3 Y36 F98 and Q100 (red sticks) show the biggest conformational differences between dimer interfaces.

A Conversation with P. James E. Peebles
P. James E. Peebles Albert Einstein Professor of Science Emeritus at Princeton University talks about his life and career with Sandra Faber University Professor of Astronomy and Astrophysics at the University of California Santa Cruz and Editor of the Annual Review of Astronomy and Astrophysics. Dr. Peebles describes his youth in Winnipeg Canada. He began his studies at the University of Manitoba where he entered the engineering program eventually transferring to Physics. On his advisor Ken Standing's urging Dr. Peebles moved to Princeton University and joined the working group of Robert H. Dicke in which he studied gravity physics. Dr. Peebles went on to develop the field of physical cosmology and his work contributed to establishing the big bang model and furthering our understanding of dark matter dark energy and the theory of structure formation.
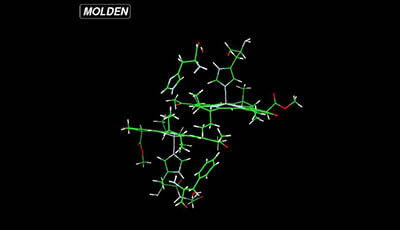
Solid-State NMR of Nanomachines Involved in Photosynthetic Energy Conversion: Supplemental Video 1
A supplemental video from the 2013 review by A. Alia et al. "Solid-State NMR of Nanomachines Involved in Photosynthetic Energy Conversion" from the Annual Review of Biophysics.
Shows the normal mode at about 50 cm-1 which has a large projection on the hydrogen bond between HisL168 and the 31 acetyl group of PL. This mode is strongly coupled to the PL HOMO energy and thus drives a dynamical localization of the HOMO and tunes the biophysical properties of the special pair.
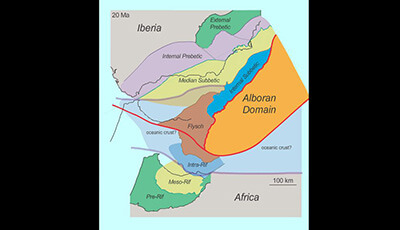
The Betic-Rif Arc and Its Orogenic Hinterland: A Review: Supplemental Video 1

A Conversation with Marc Van Montagu
Marc Van Montagu President of the European Federation of Biotechnology (EFB) and former Professor of Molecular Biology at the University of Ghent talks about his life and career with Joanne Chory Professor of Plant Molecular and Cellular Biology at the Salk Institute for Biological Studies. Dr. Van Montagu recounts how he went from studying chemistry to discovering the gene transfer mechanism from Agrobacterium to plants which opened the door to gene engineering and the creation of transgenic plants. Through the EFB and the Institute of Plant Biotechnology Outreach of which he is the founder and chairman Dr. Montagu is now dedicated to educating the general public and informing political leaders about the necessity of using science and plant engineering to prepare a sustainable future for the planet and its growing population.

A Conversation with Pierre Joliot-Curie
Pierre Joliot-Curie Professor of Biology at the Collège de France and former Director of the CNRS (National Center for Scientific Research) talks about his life and career with Jean-David Rochaix Professor of Molecular Biology at the University of Geneva. Dr. Joliot-Curie whose grandparents Pierre and Marie Curie and parents Frédéric Joliot and Irène Curie were all Nobel Prize laureates discusses his love of research which he equates to an "artistic" endeavor and an adventure. He recounts his beginnings growing up as a "poor student" in a scientific family and how his father encouraged him to study biology rather than physics. He also explains how he came to develop instruments to study his chosen subject photosynthesis. As a mentor Dr. Joliot-Curie encouraged younger generations of researchers to have fun with their work by giving them large degrees of freedom.
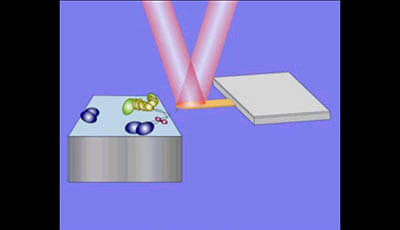
High-Speed AFM and Applications to Biomolecular Systems: Supplemental Video 1
A supplemental video from the 2013 review by Toshio Ando Takayuki Uchihashi and Noriyuki Kodera "High-Speed AFM and Applications to Biomolecular Systems" from the Annual Review of Biophysics.
Principle of tapping mode AFM. The cantilever is oscillated in the z-direction at its resonant frequency. The position of the laser beam reflected back from the cantilever is monitored to measure the cantilever deflection. The oscillation amplitude reduced by tip tapping on the sample surface is maintained constant by moving the sample stage in the z-direction through feedback control. The resulting sample stage movement traces the sample surface. Therefore the sample surface topography can be reconstructed by the use of a signal by which the z-scanner is driven.
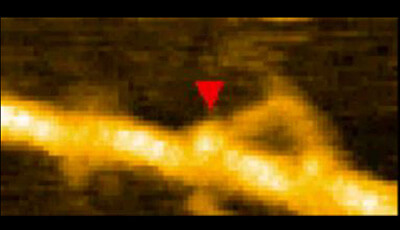
High-Speed AFM and Applications to Biomolecular Systems: Supplemental Video 2
A supplemental video from the 2013 review by Toshio Ando Takayuki Uchihashi and Noriyuki Kodera "High-Speed AFM and Applications to Biomolecular Systems" from the Annual Review of Biophysics.
Processive movement of myosin V (M5-HMM). The dynamic process in 1 µM ATP was captured at 7 fps. Scan range 130 × 65 nm2 with 80 × 40 pixel. Foot stomp event occurred at the trailing head is marked with a red triangle.

High-Speed AFM and Applications to Biomolecular Systems: Supplemental Video 3
A supplemental video from the 2013 review by Toshio Ando Takayuki Uchihashi and Noriyuki Kodera "High-Speed AFM and Applications to Biomolecular Systems" from the Annual Review of Biophysics.
Hand-over-hand movement of myosin V (M5-HMM) including foot stomp of the leading head. The dynamic process in 1 µM ATP was captured at 7 fps. Scan range 150 × 75 nm2 with 80 × 40 pixel. Five frames are selected and shown in Figure 4c. Foot stomp events occurred at the leading head are marked with light-blue triangles.

High-Speed AFM and Applications to Biomolecular Systems: Supplemental Video 4
A supplemental video from the 2013 review by Toshio Ando Takayuki Uchihashi and Noriyuki Kodera "High-Speed AFM and Applications to Biomolecular Systems" from the Annual Review of Biophysics.
Long tracking of myosin V (M5-HMM) walking along actin filament. This typical movie showing long processive runs in 1 µM ATP was captured at 7 fps. To chase the M5-HMM molecule the scan area was moved. Scan range 150 × 75 nm2 with 80 × 40 pixel; the whole imaging area 560 × 120 nm2; number of steps observed 14. Foot stomp events at the leading head are marked with light-blue triangles.
