Multimedia
Filter :
Publication date
Article type
Subject

Molecular Mechanisms by Which Marine Phytoplankton Respond to Their Dynamic Chemical Environment: Video 1
A video from the 2015 review by Brian Palenik "Molecular Mechanisms by Which Marine Phytoplankton Respond to Their Dynamic Chemical Environment" from the Annual Review of Marine Science.
Shown: Time-lapse image series of light production from two Synechococcus elongatus strains that carry a gene encoding the firefly luciferase reporter enzyme. The strains were inoculated onto agar plates in the shape of a moon and sun and their luciferase genes are driven by different regulatory DNA elements that cause gene activity to peak with opposite phases approximately 12 h apart. Video created by Shannon Mackey Mark Zoran and Susan Golden at the Department of Biology Texas A&M University and provided courtesy of Susan Golden Center for Chronobiology University of California San Diego.

Gecko Adhesion as a Model System for Integrative Biology, Interdisciplinary Science, and Bioinspired Engineering: Video 1
A video from the 2014 review by Kellar Autumn Peter Niewiarowski and Jonathan B. Puthoff "Gecko Adhesion as a Model System for Integrative Biology Interdisciplinary Science and Bioinspired Engineering" from the Annual Review of Ecology Evolution and Systematics.
Shown: Zoom into the tokay gecko’s adhesive system from the macro- to nanoscales. For information on the helium ion scanning electron microscopy technique used in this video see Yang et al. (2011).
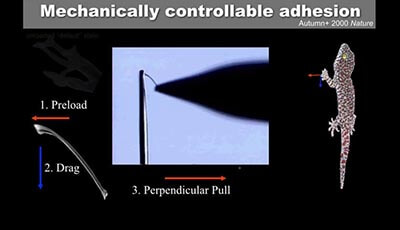
Gecko Adhesion as a Model System for Integrative Biology, Interdisciplinary Science, and Bioinspired Engineering: Video 2
A video from the 2014 review by Kellar Autumn Peter Niewiarowski and Jonathan B. Puthoff "Gecko Adhesion as a Model System for Integrative Biology Interdisciplinary Science and Bioinspired Engineering" from the Annual Review of Ecology Evolution and Systematics.
Video and animation illustrating the mechanical requirements for attachment and detachment of a single isolated gecko seta (Autumn et al. 2000). The video shows a single seta glued to a minutien pin. The vertical bar at the left side is a 25-μm aluminum wire force gauge. Initial attempts to adhere a single isolated seta to a surface failed because we simply touched the tip of the seta into the surface and pulled away vertically. Instead a slight preload force followed by a micrometer-scale drag along the direction of curvature of the seta (i.e. toward the rear of the animal) switches the spatulae from their default unloaded state to the adhered state. The seta can now sustain a perpendicular pull because the adhesive van der Waals forces at the spatula tips resist detachment. Detachment occurs when the angle between the setal shaft and the surface exceeds 30°. This experiment illustrates the mechanical program for attachment and detachment required for controllable adhesion in gecko setae.

Gecko Adhesion as a Model System for Integrative Biology, Interdisciplinary Science, and Bioinspired Engineering: Video 3
A video from the 2014 review by Kellar Autumn Peter Niewiarowski and Jonathan B. Puthoff "Gecko Adhesion as a Model System for Integrative Biology Interdisciplinary Science and Bioinspired Engineering" from the Annual Review of Ecology Evolution and Systematics.
Toe peeling (digital hyperextension) during climbing by a tokay gecko. The motion of gecko toes is superficially similar to that of peeling tape. However because adhesion of gecko toes is governed by the micro-mechanics of their setae a tape peeling model can be rejected (Autumn et al. 2006a). In contrast to the peeling of tape gecko toes function by “frictional adhesion”: Pull-off forces increase linearly with shear load and detach when the angle of the resultant force exceeds 30° relative to the surface.
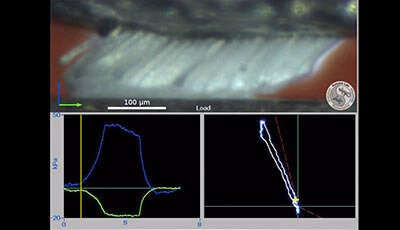
Gecko Adhesion as a Model System for Integrative Biology, Interdisciplinary Science, and Bioinspired Engineering: Video 4
A video from the 2014 review by Kellar Autumn Peter Niewiarowski and Jonathan B. Puthoff "Gecko Adhesion as a Model System for Integrative Biology Interdisciplinary Science and Bioinspired Engineering" from the Annual Review of Ecology Evolution and Systematics.
Video 4 shows how gecko setae are slippery not sticky when pushed away from the animal against the direction of curvature of the setae (this is the opposite direction geckos use when they climb). In the lower left a compression force (blue line) develops during the load step. Friction (shear force) is approximately 0.25 of the compression force as expected for conventional friction of hard dry materials in contact. There is no measurable adhesion when setae are pushed against their curvature: This is the anti-adhesive direction.
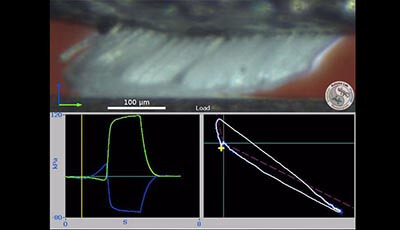
Gecko Adhesion as a Model System for Integrative Biology, Interdisciplinary Science, and Bioinspired Engineering: Video 5
A video from the 2014 review by Kellar Autumn Peter Niewiarowski and Jonathan B. Puthoff "Gecko Adhesion as a Model System for Integrative Biology Interdisciplinary Science and Bioinspired Engineering" from the Annual Review of Ecology Evolution and Systematics.
Video 5 shows how gecko setae adhere when preloaded and dragged along the direction of curvature of the setae (this is in the same direction geckos use when they climb). In the lower left the normal force (blue line) is compressive initially during the load step but immediately following the drag step strong adhesion occurs (negative normal force) and the setae are drawn into tension. Notably adhesion is sustained even as the setae slide across the substrate. The lower right plot illustrates frictional adhesion: Adhesion is coupled to friction and the resultant force angle is approximately 30°. Adhesion is controlled by the shear force. During the pull step shear force decreases and adhesion returns to zero without the tacky behavior common to conventional adhesive tapes.
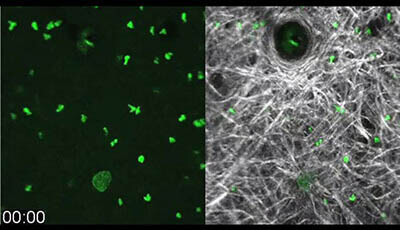
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 1
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: Biphasic neutrophil swarming response to focal tissue damage
Bone marrow-derived neutrophils from C57BL/6 mice were CMFDA-labeled and injected intradermally into the ventral ear skin of a Tyrc-2J/c-2J mouse 3 h before laser-induced focal tissue damage. This representative video shows the biphasic chemotactic response of neutrophils (pseudo-colored in green) sensing the focal tissue damage (autofluorescence green) (left) within the fibrous collagenous connective tissue of the ear skin dermis (visualized by second harmonic generation white) (right).
Original publication: Lämmermann T Afonso PV Angermann BR Wang JM Kastenmuller W et al. 2013. Neutrophil swarms require LTB4 and integrins at sites of cell death in vivo. Nature 498:371–75
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 2
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: Congregating neutrophils displace fibrous collagen bundles from the wound center
This video shows the collagenous fiber network (visualized by second harmonic generation) as neutrophils accumulate at the focal damage site in the dermis of a LysMgfp/+Tyrc-2J/c-2J mouse. Fiber bundles are displaced over time in the x-y axis. Some disappearing fibers are also displaced in the z-axis out of the imaging volume (not shown).
Original publication: Lämmermann T Afonso PV Angermann BR Wang JM Kastenmuller W et al. 2013. Neutrophil swarms require LTB4 and integrins at sites of cell death in vivo. Nature 498:371–75
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 3
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: (a) Rapid viral gene expression in SCS myeloid cells after MVA infection
Popliteal LNs from MVA NP-S-GFP(nuclear)-infected (footpad) animals were surgically exposed and analyzed by two-photon microscopy. Video shows onset of virally driven GFP expression in situ and identifies CD169+ myeloid cells as viral targets.
(b) Virally infected CD169+ myeloid cells actively move their fine processes
Popliteal LNs from MVA GFP(cytosolic)-infected (footpad) animals were surgically exposed and analyzed by two-photon microscopy. Video shows dynamic movement of infected GFP-expressing myeloid cells in situ.
Original publication: Kastenmüller W Brandes M Wang Z Herz J Egen JG Germain RN. 2013. Peripheral prepositioning and local CXCL9 chemokine-mediated guidance orchestrate rapid memory CD8+ T cell responses in the lymph node. Immunity 38:502–13
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 4
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: CXCR6-GFPbright cells are prepositioned near the LN capsule
A total of 3 × 106 CMTMR labeled naïve OT-1 T cells (red) were transferred into a CXCR6gfp/+ recipient. 24 h later the popliteal LN was surgically exposed and imaged using a two-photon microscope. The video shows a z stack (250 µm) scanned from the capsule to the paracortex of the LN.
Original publication: Kastenmüller W Torabi-Parizi P Subramanian N Lammermann T Germain RN. 2012. A spatially-organized multicellular innate immune response in lymph nodes limits systemic pathogen spread. Cell 150:1235–48

Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 5
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: CXCR6-GFPbright (NKT) cells are highly motile
Popliteal LNs from CXCR6GFP/+ /CD11cYFP/+ mice were surgically exposed and analyzed by two-photon microscopy. Video shows dynamic behavior of CXCR6-GFPbright cells close to the LN capsule near the interfollicular area.
Original publication: Kastenmüller W Torabi-Parizi P Subramanian N Lammermann T Germain RN. 2012. A spatially-organized multicellular innate immune response in lymph nodes limits systemic pathogen spread. Cell 150:1235–48
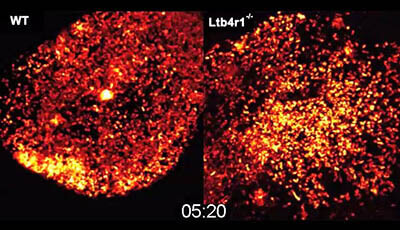
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 6
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
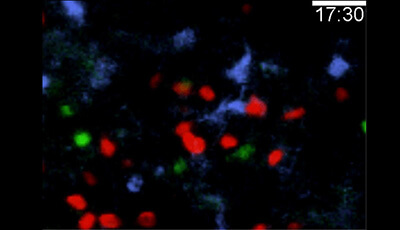
Shown: LTB4 requirement for swarming in infected lymph nodes
Mice were infected with P. aeruginosa-GFP in the footpad before 2P-IVM was performed on the draining popliteal lymph nodes when comparable neutrophil numbers were present in the subcapsular sinus at indicated times after infection (WT: 3 h Ltb4r1−/−: 4.5 h). Neutrophil-GFP signal is pseudo-colored (heat map) to indicate neutrophil clusters (white) in WT-LysMgfp/+ mice (left) and Ltb4r1−/−LysMgfp/+ mice (right).
Original publication: Lämmermann T Afonso PV Angermann BR Wang JM Kastenmuller W et al. 2013. Neutrophil swarms require LTB4 and integrins at sites of cell death in vivo. Nature 498:371–75
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 7
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
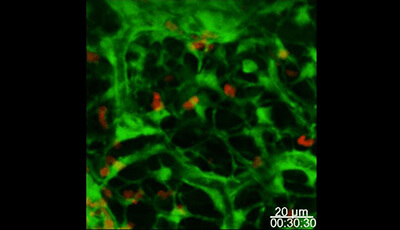
Shown: Dynamic behavior of naïve CD4+ and CD8+ T cells in the lymph node
CD4+ T cells (red) and CD8+ T cells (green) in the inguinal LN (Mandl JN Liou R Klauschen F Vrisekoop N Monteiro JP et al. 2012. Quantification of lymph node transit times reveals differences in antigen surveillance strategies of naive CD4+ and CD8+ T cells. Proc. Natl. Acad. Sci. USA 109:18036–41).
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 8
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
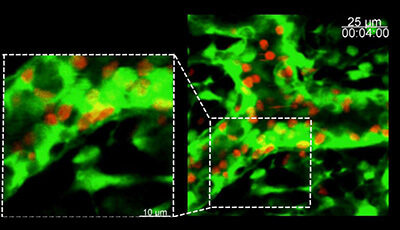
Shown: T cells migrate along the FRC network
Dynamic image of T cell (red) migration along the FRC network (green). The trails of three of the T cells are highlighted in the second part of the video with colored dots to help visualize the path taken along the fibers by a given T cell.
Original publication: Bajenoff M Egen JG Koo LY Laugier JP Brau F et al. 2006. Stromal cell networks regulate lymphocyte entry migration and territoriality in lymph nodes. Immunity 25:989–1001
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 9
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
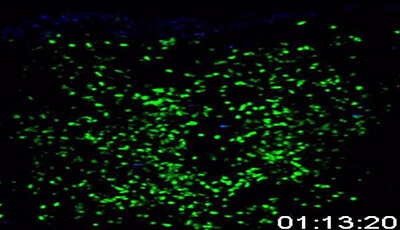
Shown: T cells exit HEVs in limited locations
A single z slice from an intravital 4D data set showing numerous T cells exiting HEV via lucent areas that appear to be gaps in the FRC sheath ("exit ramps").
Original publication: Bajenoff M Egen JG Koo LY Laugier JP Brau F et al. 2006. Stromal cell networks regulate lymphocyte entry migration and territoriality in lymph nodes. Immunity 25:989–1001
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 10
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: (a) Kinetics of naive CD8+ T cell priming
Popliteal LNs from MVA OVA-infected (footpad) animals were surgically exposed and analyzed by two-photon microscopy. Prior (24 h) to infection mice received labeled (CMFDA) naive (CD44lo) OT-I T cells. Video shows the dynamic behavior and cluster formation of OT-I T cells in the course of infection as well as the positioning of the clusters in relation to HEV.
(b) Migrational arrest of antigen-specific CD8+ T cells after infection
Popliteal LNs from MVA OVA-infected (footpad) animals were surgically exposed and analyzed by two-photon microscopy. Prior (24 h) to infection mice received labeled (CMFDA) naive (CD44lo) OT-I T cells and labeled (CTR) naive (CD44lo) polyclonal control T cells. Video shows migrational arrest of OT-I and active migration of nonspecific polyclonal CD8+ T cells.
Original publication: Kastenmuller W Brandes M Wang Z Herz J Egen JG Germain RN. 2013. Peripheral prepositioning and local CXCL9 chemokine-mediated guidance orchestrate rapid memory CD8+ T cell responses in the lymph node. Immunity 38:502–13
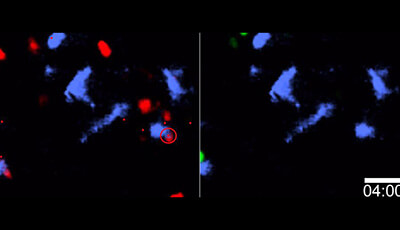
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 11
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: CCR5 guides naive CD8+ T cells to helped DC
Video illustrates differences in the contact frequency between CD4+ T cell helped (blue) DCs and each of the two polyclonal CD8+ T cell populations. 57 contacts are made between CD4+ T cell helped DCs and WT CD8+ T cells (red dots and circles left panel) as compared to 6 contacts between the same DCs and CCR5 −/− CD8+ T cells (green dots and circles right panel) resulting in a calculated hit rate ratio of 3.26 for WT versus CCR5 −/− CD8+ T cells interacting with DCs.
Original publication: Castellino F Huang AY Altan-Bonnet G Stoll S Scheinecker C Germain RN. 2006. Chemokines enhance immunity by guiding naive CD8+ T cells to sites of CD4+ T cell-dendritic cell interaction. Nature 440:890–95
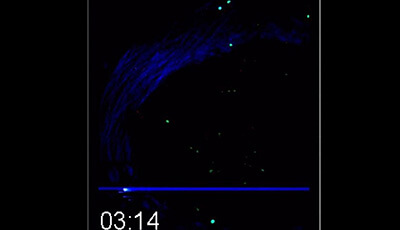
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 12
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: (a) Recruitment of antigen-unspecific memory CD8+ T cells to the SCS area
Popliteal LNs from MVA WT-infected (footpad) mice harboring memory OT-I T cells were surgically exposed and analyzed by two-photon microscopy. Video shows recruitment to the SCS and swarming of memory OT-I T cells in the absence of cognate antigen.
(b) Recruitment of antigen-unspecific memory CD8+ T cells to the SCS area depends on CXCR3
Popliteal LNs from CD11cYFP mice infected with MVA WT (footpad) harboring WT and Cxcr3 −/− memory OT-I T cells were surgically exposed and analyzed by two-photon microscopy. Video shows differential migration patterns of WT versus Cxcr3 −/− memory OT-I T cells and swarming behavior of WT but not Cxcr3 −/− memory OT-I T cells around DCs.
Original publication: Kastenmüller W Brandes M Wang Z Herz J Egen JG Germain RN. 2013. Peripheral prepositioning and local CXCL9 chemokine-mediated guidance orchestrate rapid memory CD8+ T cell responses in the lymph node. Immunity 38:502–13
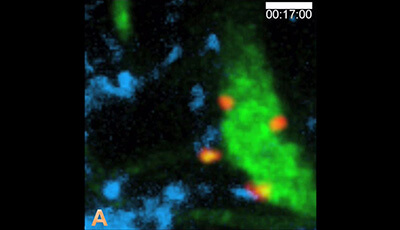
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 13
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: The calcium responses of immigrant Ag-specific B cells engaging Ag-bearing DCs immediately after exiting HEVs
(a) Immediately after exiting a HEV an MD4 B cell (red pointed by an arrowhead while still inside the HEV) engages an HEL-DC (blue) and shows an abrupt increase in Fluo-4 fluorescence indicating elevation of intracellular [Ca2+]. (b) The same video is played again showing magnified views (2×) including freeze frames to highlight the first DC contact and the subsequent onset of calcium flux by the MD4 cell. Scale bar: 20 µm.
Original publication: Qi H Egen JG Huang AY Germain RN. 2006. Extrafollicular activation of lymph node B cells by antigen-bearing dendritic cells. Science 312:1672–76
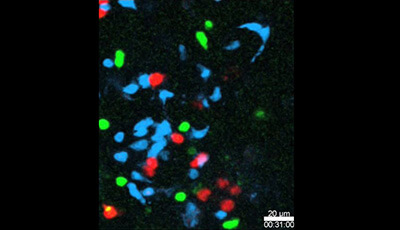
Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue: Video 14
A video from the 2014 review by Hai Qi Wolfgang Kastenmüller and Ronald N. Germain "Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue" from the Annual Review of Cell and Developmental Biology.
Shown: Sap −/− T cells fail to stably interact with B cells when examined together with Sap +/+T cells in the same LN in vivo
Sap +/+ OT-2 T cells (red) Sap −/− OT-2 T cells (green) and MD4 B cells (blue) were cotransferred into mice that had previously been immunized with HEL-OVA conjugate antigen. Intravital imaging of the draining LNs was performed 24 to 36 h after cell transfer. The circle at the beginning of this video highlights a three-cell cluster in which an MD4 B cell is engaged by both a Sap +/+ and a Sap −/− OT-2 T cell. The Sap −/− T cell then disengages quickly while the Sap +/+ T cell remains in conjugation with the B cell for a much longer period of time.
Original publication: Qi H Cannons JL Klauschen F Schwartzberg PL Germain RN. 2008. SAP-controlled T-B cell interactions underlie germinal centre formation. Nature 455:764–69
